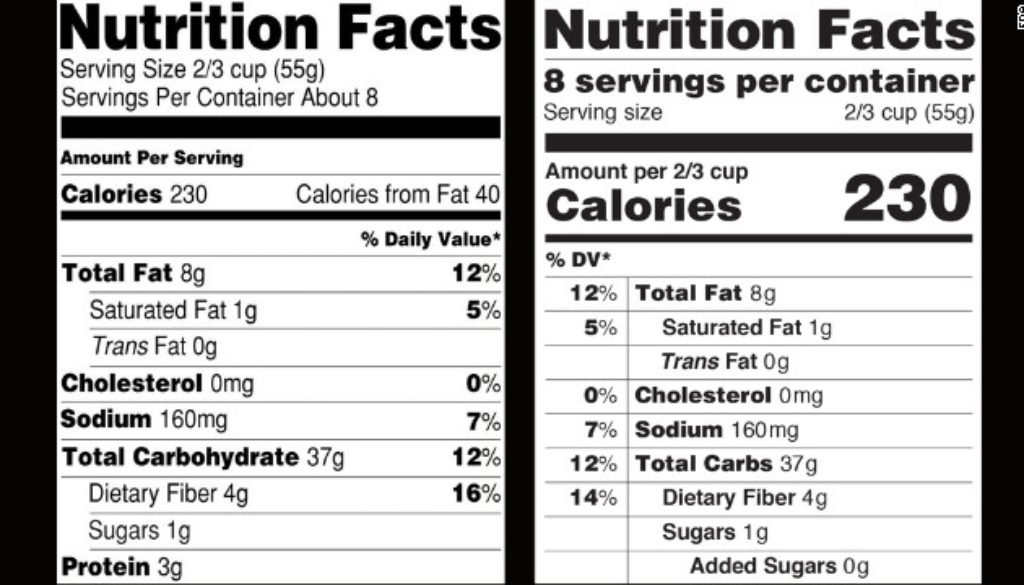
Pointless Delay to the Added Sugar Label Keeps Consumers in the Dark
By Genna Reed, Union of Concerned Scientists
In another frustrating example of undermining science-based protections, the FDA this morning proposed delaying compliance for revisions to the Nutrition Facts label.
Most food companies were supposed to roll out their revised labels by July 2018. This delay would mean that those initial, larger companies would have until January 2020 and smaller companies until January 2021.
I have been dreading this official announcement all year and hoping—as more and more products I see in stores have updated their labels—that the FDA would acknowledge that its original rule was perfectly reasonable and has already given companies ample time to comply.
In December, food industry leaders proposed two different riders to draft House appropriations legislation that would have delayed the rule. Luckily, those failed to make it into final language.
Then, in April at now-FDA Commissioner Scott Gottlieb’s confirmation hearing, he implied that he might delay the revised nutrition facts label. I urged Gottlieb to keep the compliance dates as a part of the final rule that was issued in 2016.
Once confirmed, Gottlieb was faced with what I would consider a pretty clear-cut decision: Implement a rule that was based in clear science on the public health consequences associated with excessive added sugar consumption—one that was also supported by the expert-driven Dietary Guidelines recommendations—or cow to industry wishes to delay the rule, even though the majority of food companies would have had until 2019 to make the new changes to their labels, and larger food companies like Mars, Inc. and Hershey Co. have already met the deadline or are on track to meet it.
In fact, according to the Center for Science in the Public Interest, at least 8,000 products from a variety of companies already bear the new label.
A few months later, the FDA announced of its intention to push back compliance dates, but there was no formal decision or indication of how long the delay would be. I, again, urged Gottlieb not to take a step backward on food label transparency by delaying the new label.
Despite what some food companies will have you believe, they have had plenty of time to accept the science on added sugar consumption and to give consumers the information for which they’ve been clamoring. The FDA first began its work to revise the nutrition facts label in 2004, and the proposed rule which included the added sugar line was issued in 2014. Industry has had over ten years to give consumers the information they want to make informed decisions, and to acknowledge the mounting evidence that excessive sugar consumption can lead to adverse health consequences, including heart disease, obesity, diabetes, and hypertension.
Instead, as we demonstrated in a 2015 analysis of public comments on the FDA’s proposed rule, the majority of unique comments supported the rule (99 percent of whom were public health experts), while 69 percent of those opposed to the rule were from the food industry. The companies’ reasons for opposition included flimsy arguments about consumers’ ability to understand nutrition labels.
Last week, we signed onto a letter along with twenty other science, public health, and consumer organizations urging Gottlieb to let the rule move forward. As we wrote in the letter, this delay means that “an entire cycle of the Dietary Guidelines for Americans will have passed without the federal government’s premier public-health regulatory agency taking final action to implement a major recommendation of the Guidelines.”
It also means that consumers will have to continue to guess how much of the sugar in their food is added, gambling on healthy food purchasing decisions. While asking the agency to delay its labeling rules, the sugar industry seems to understand that it’s actually time to reformulate and meet consumer demand for healthier products to win consumers’ trust. A surefire way to win our trust would have been to move forward with the label, not force us to wait another year and a half for information we have the right to know.
The FDA’s failure to follow the science and listen to public health experts, including HHS staff who helped write the most recent Dietary Guidelines, is incredibly disappointing. We will be weighing in on this decision with comments that will be accepted for 30 days after October 2nd and will update you on how you can tell the FDA to rescind its rule to delay the enforcement dates for added sugar labeling.